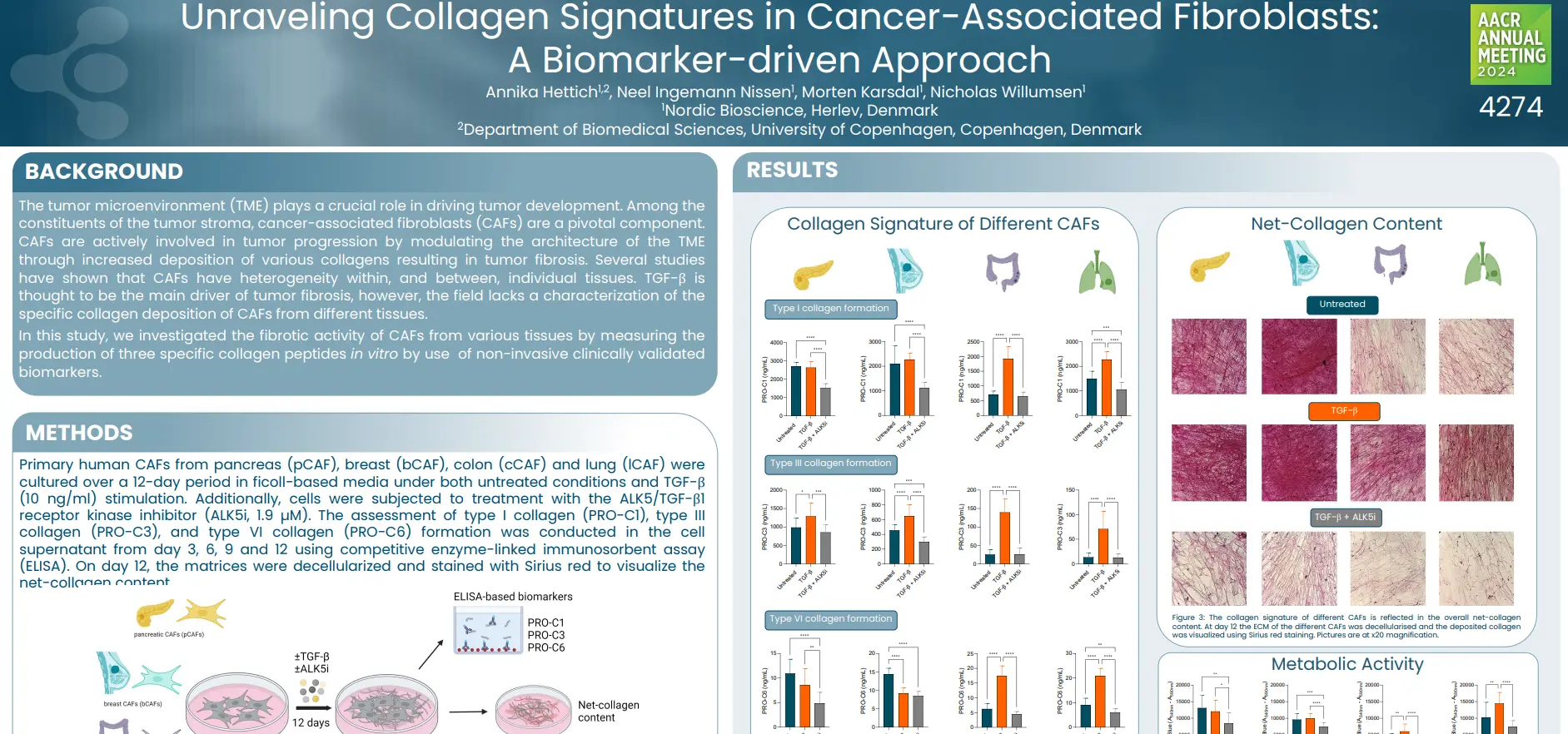
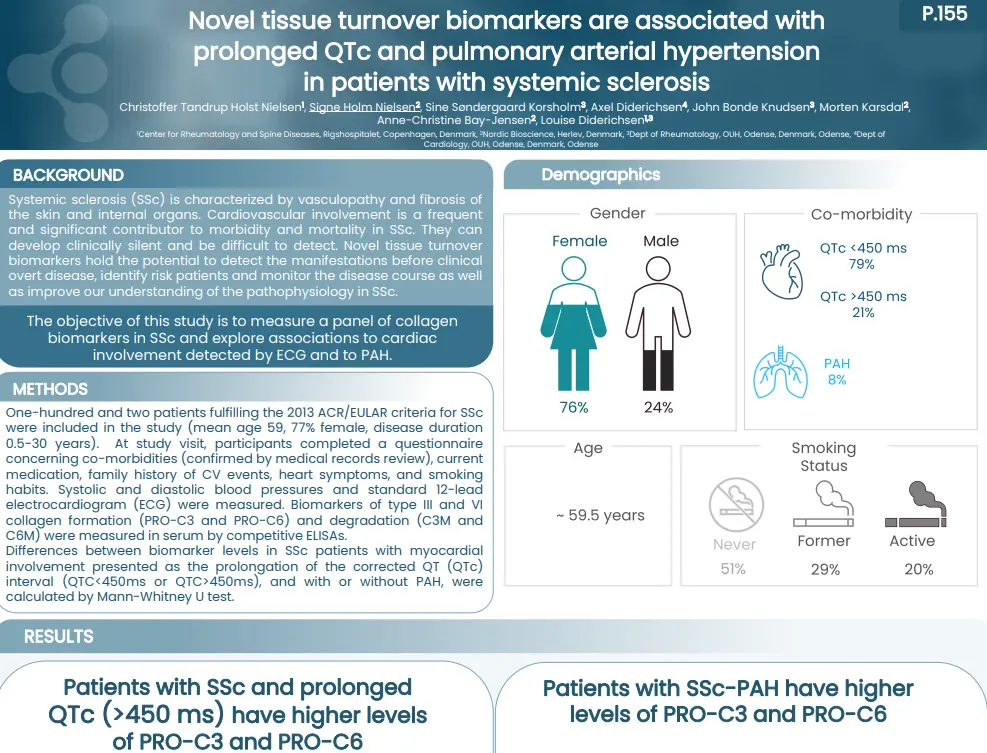
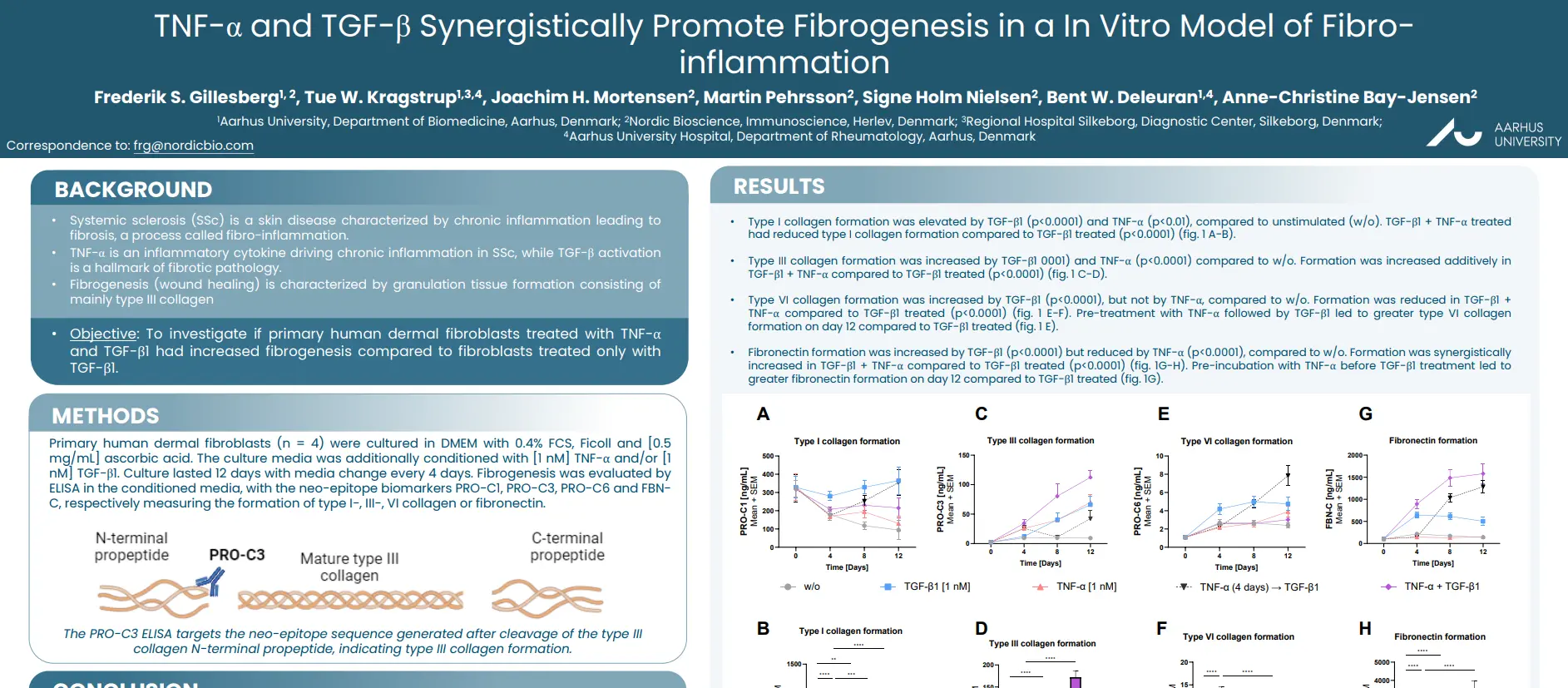
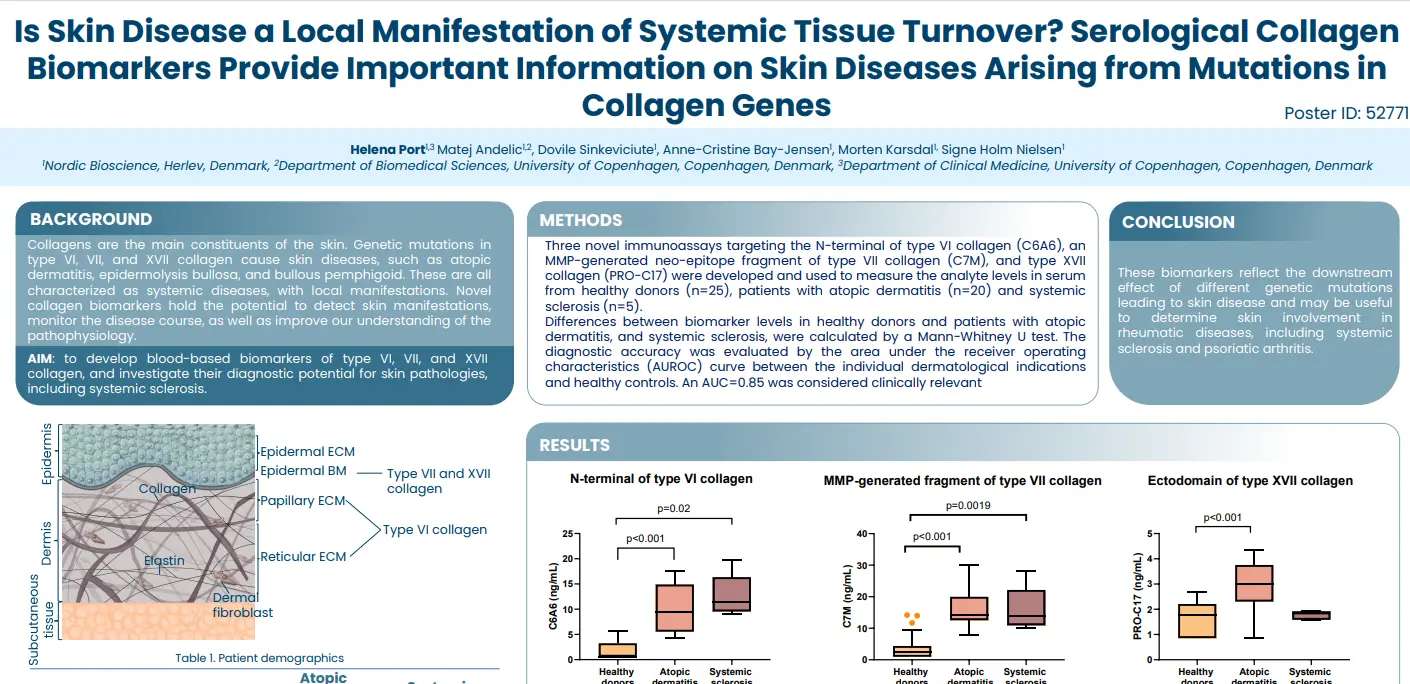
Tissue-derived peripheral biomarkers that reflect activity of T-cells, macrophages, and neutrophils in patients with solid tumors
Introduction
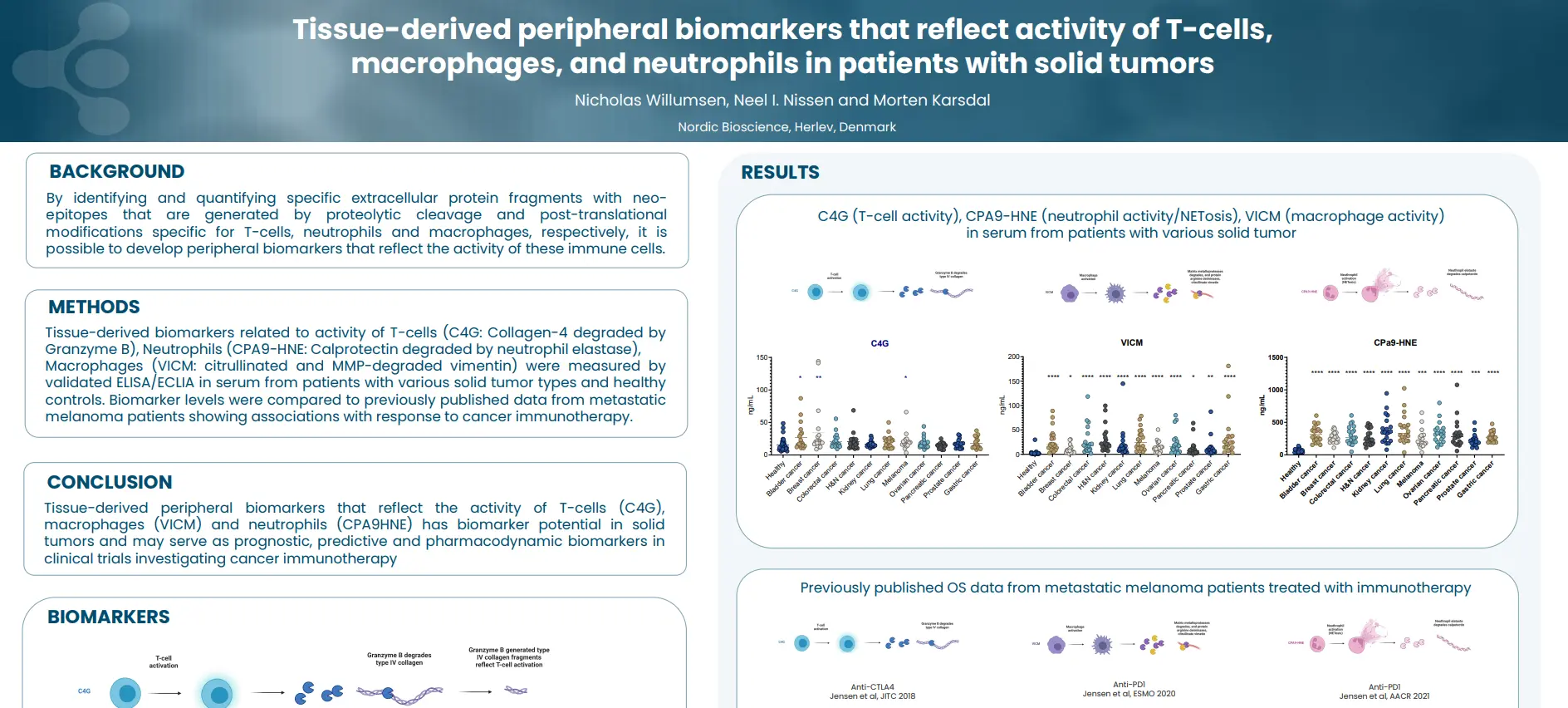
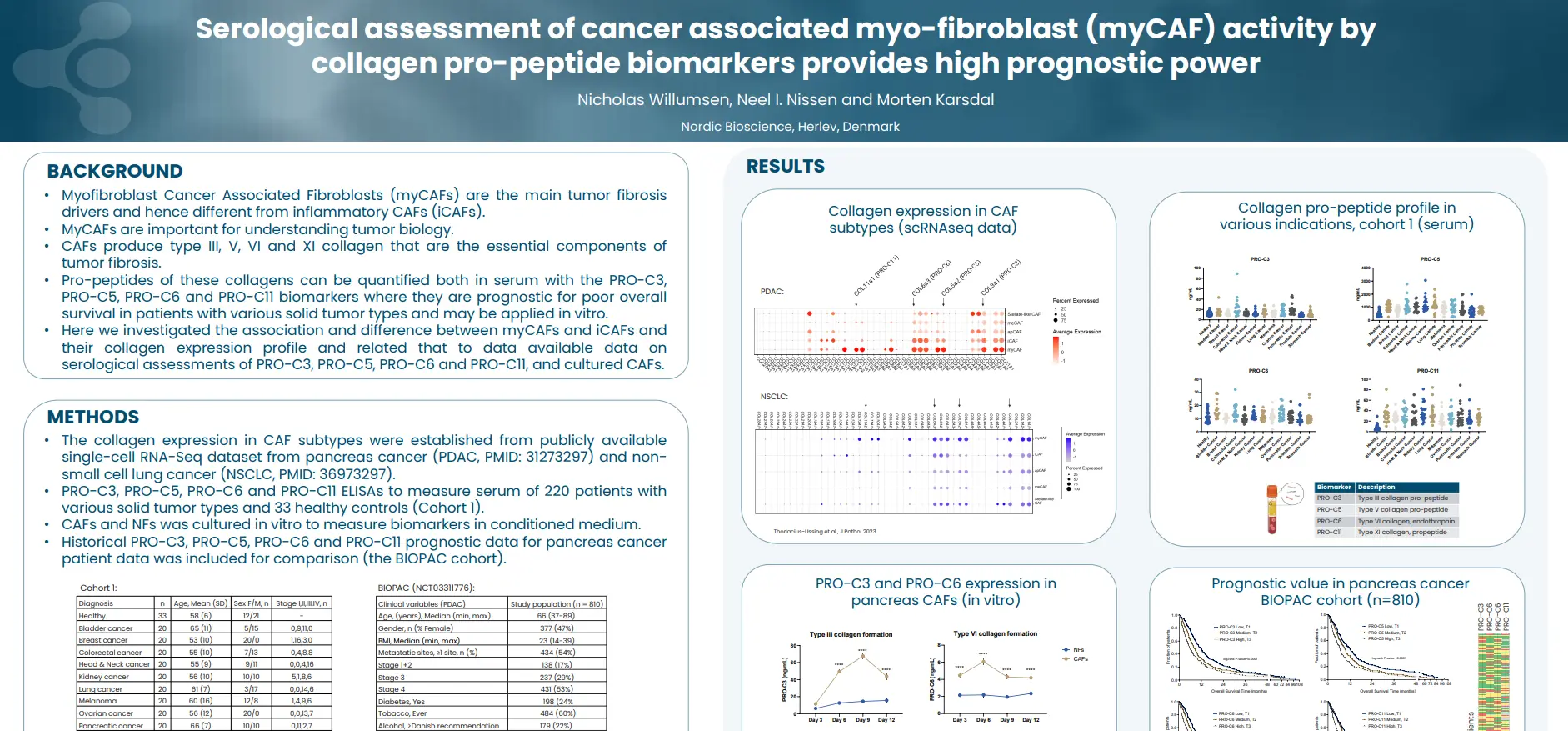
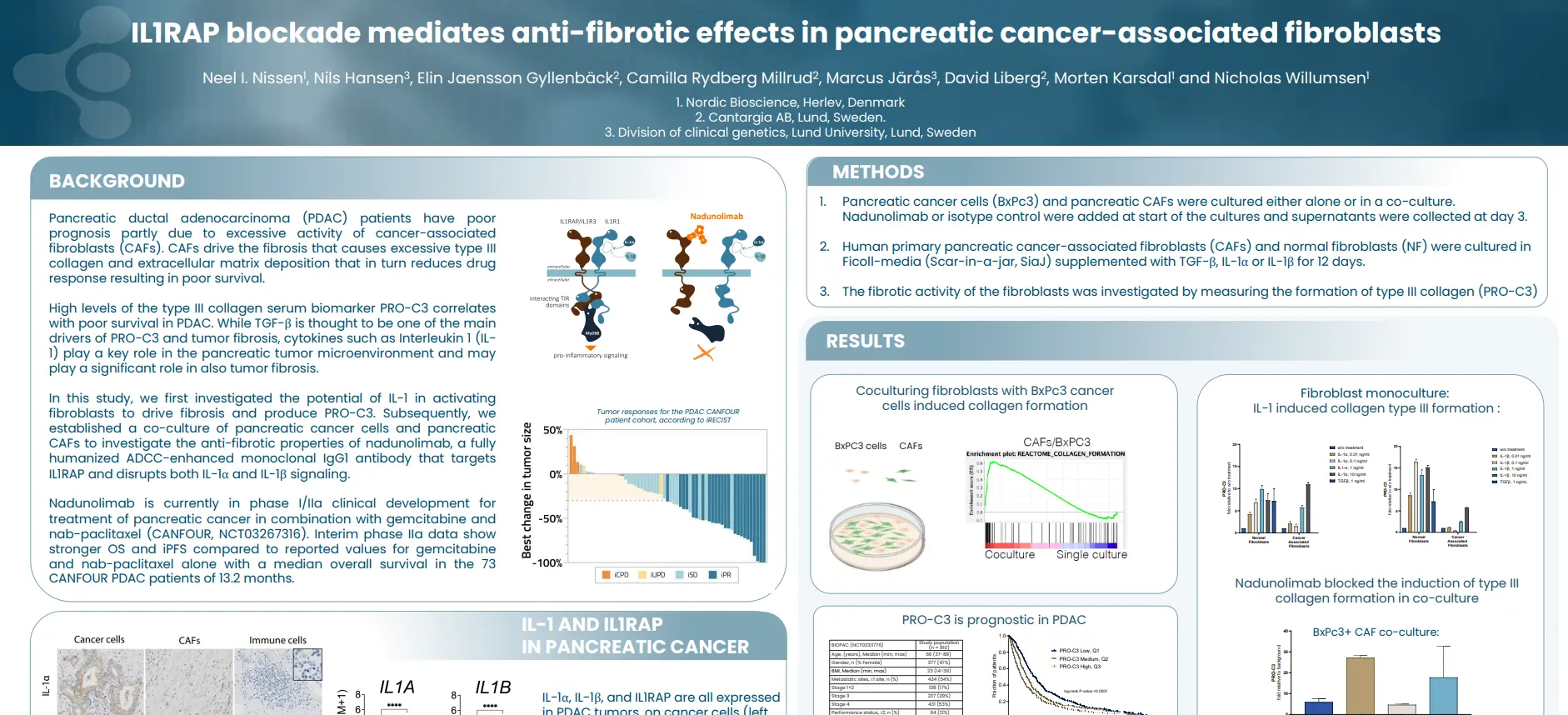
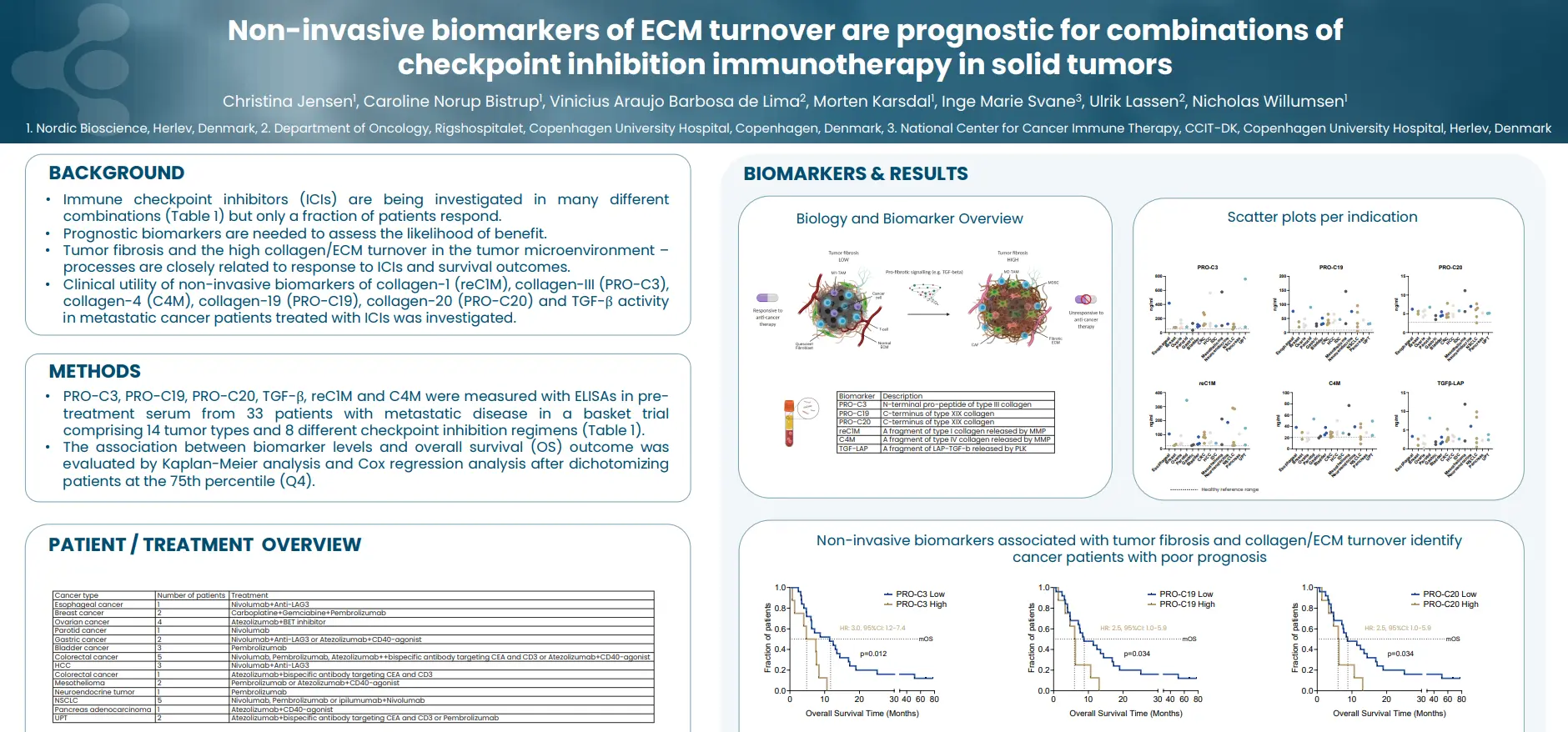
By identifying and quantifying specific extracellular protein fragments with neo-epitopes that are generated by proteolytic cleavage and post-translational modifications specific for T-cells, neutrophils and macrophages, respectively, it is possible to develop peripheral biomarkers that reflect the activity of these immune cells.
Poster
Conclusion
Tissue-derived peripheral biomarkers that reflect the activity of T-cells (C4G), macrophages (VICM) and neutrophils (nordicCPa9-HNE™) has biomarker potential in solid tumors and may serve as prognostic, predictive and pharmacodynamic biomarkers in clinical trials investigating cancer immunotherapy.