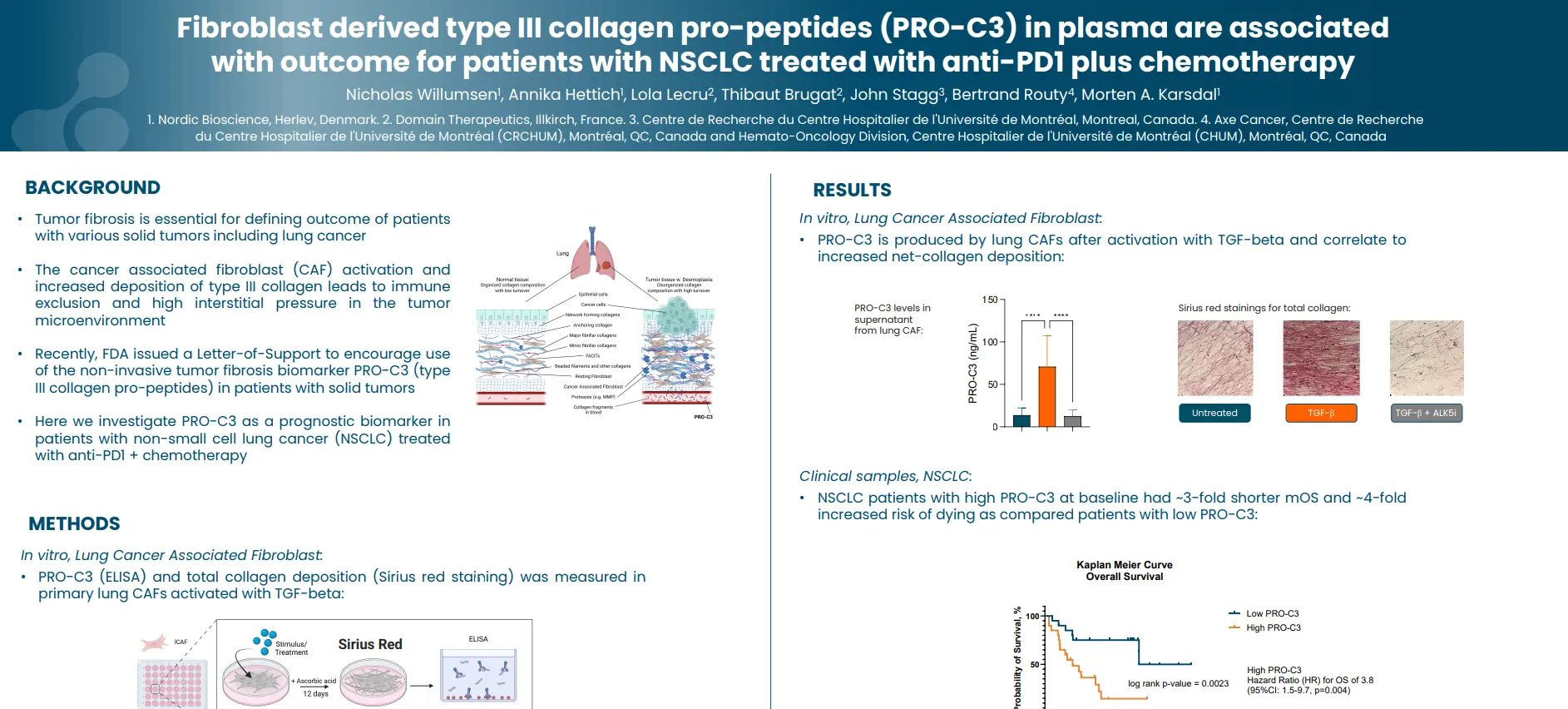
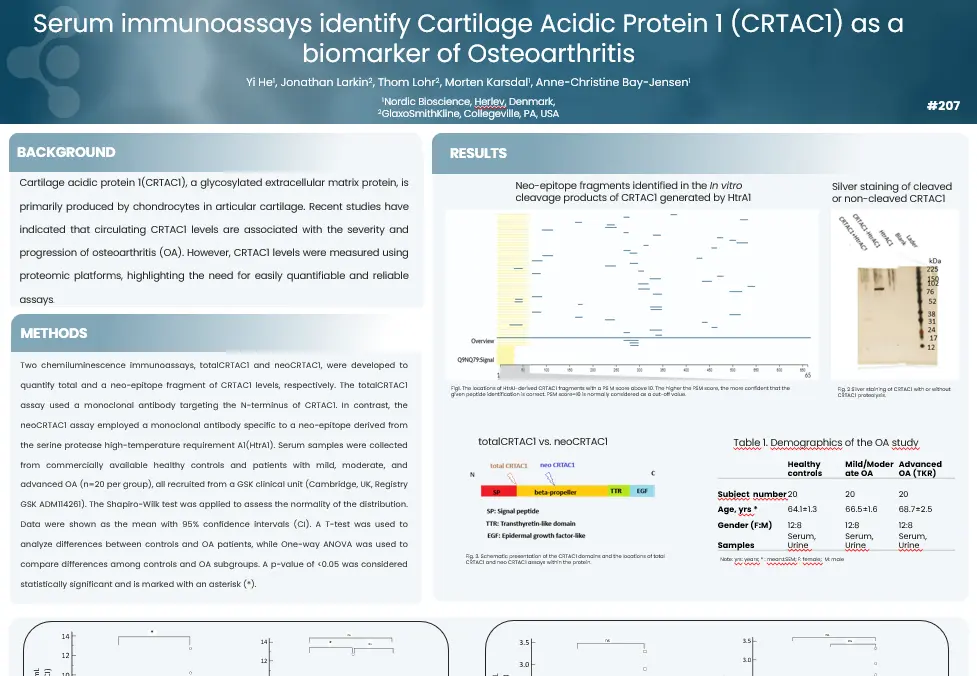
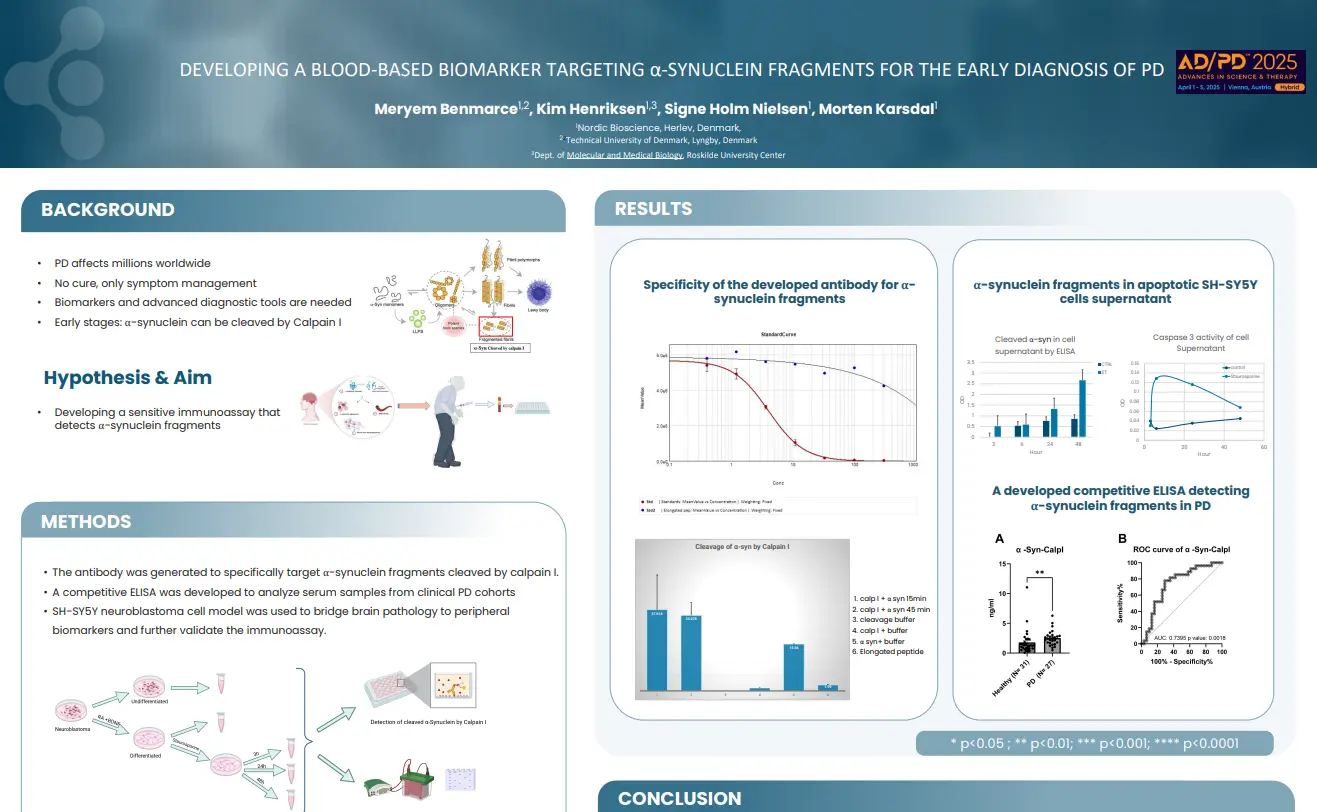
Fibroblast activation protein (FAP)- cleaved type III collagen (C3F) is a potential marker for intestinal fibrosis in patients with Crohn’s disease
Introduction
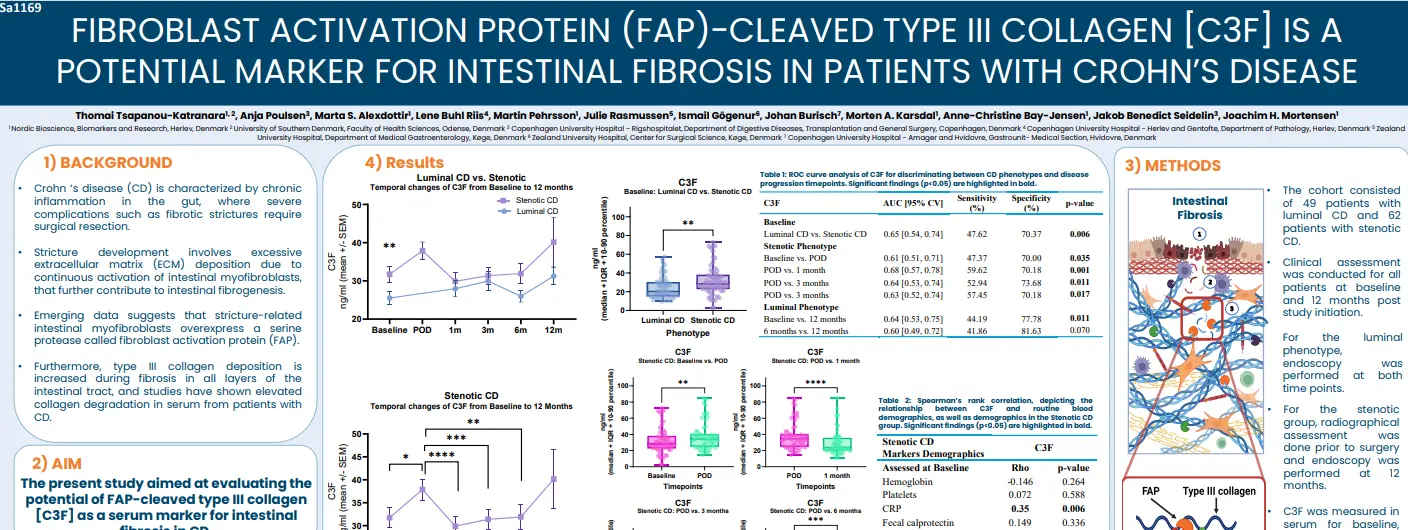
Crohn ‘s disease (CD) is characterized by chronic inflammation in the gut, where severe complications such as fibrotic strictures require surgical resection. Stricture development involves excessive extracellular matrix (ECM) deposition due to continuous activation of intestinal myofibroblasts, that further contribute to intestinal fibrogenesis. Emerging data suggests that stricture-related intestinal myofibroblasts overexpress a serine protease called fibroblast activation protein (FAP). Furthermore, type III collagen deposition is increased during fibrosis in all layers of the intestinal tract, and studies have shown elevated
collagen degradation in serum from patients with CD.
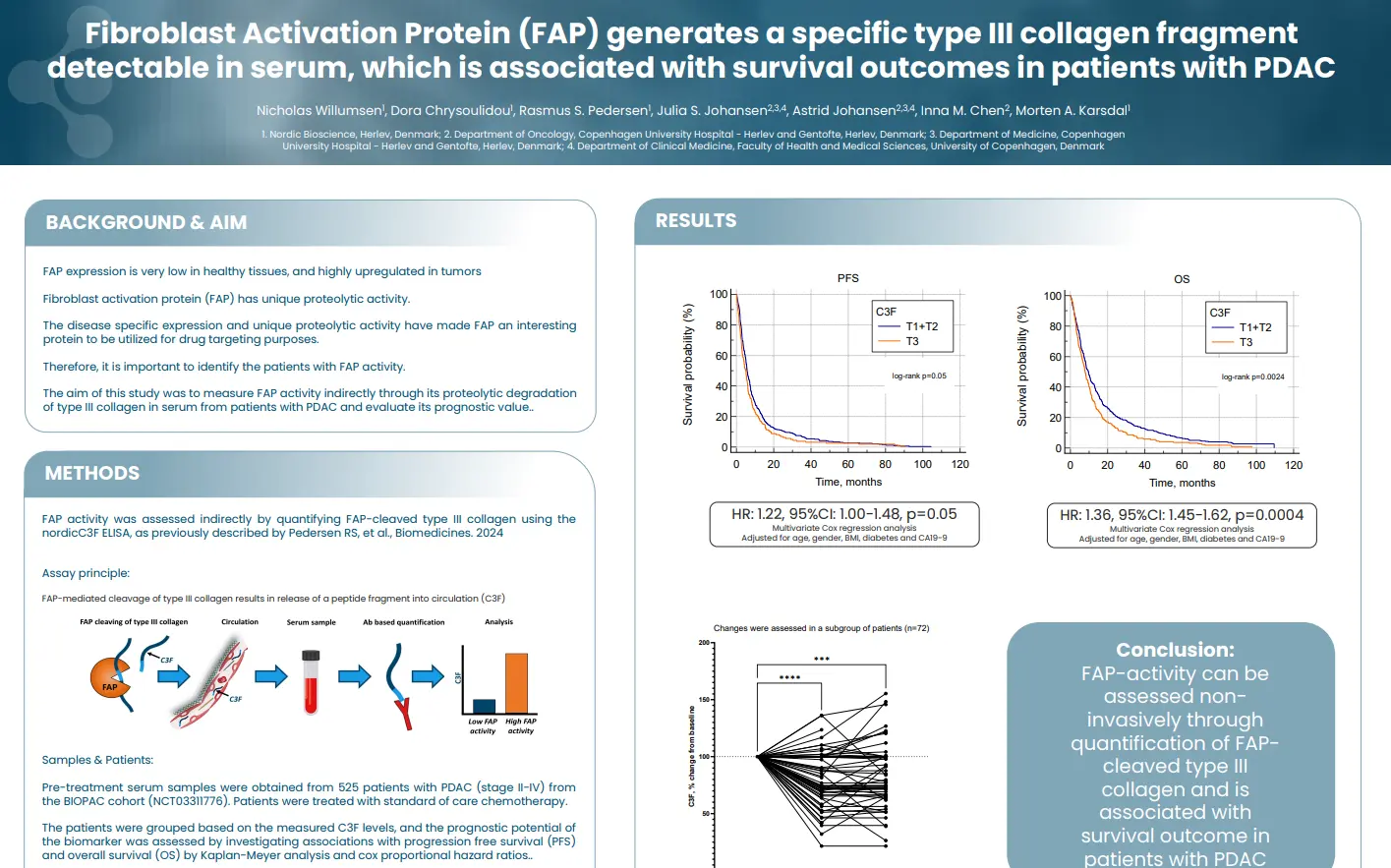
In this study we aimed to evaluate the potential of FAP-cleaved type III collagen (C3F) as a serum marker for intestinal fibrosis in CD.
Poster
Conclusion
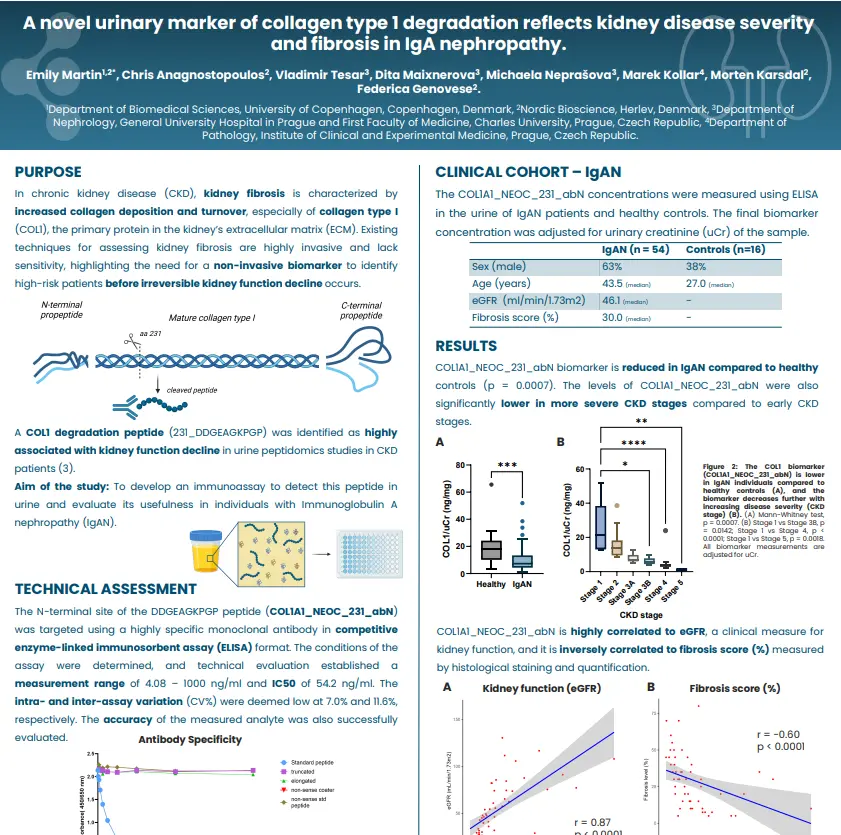
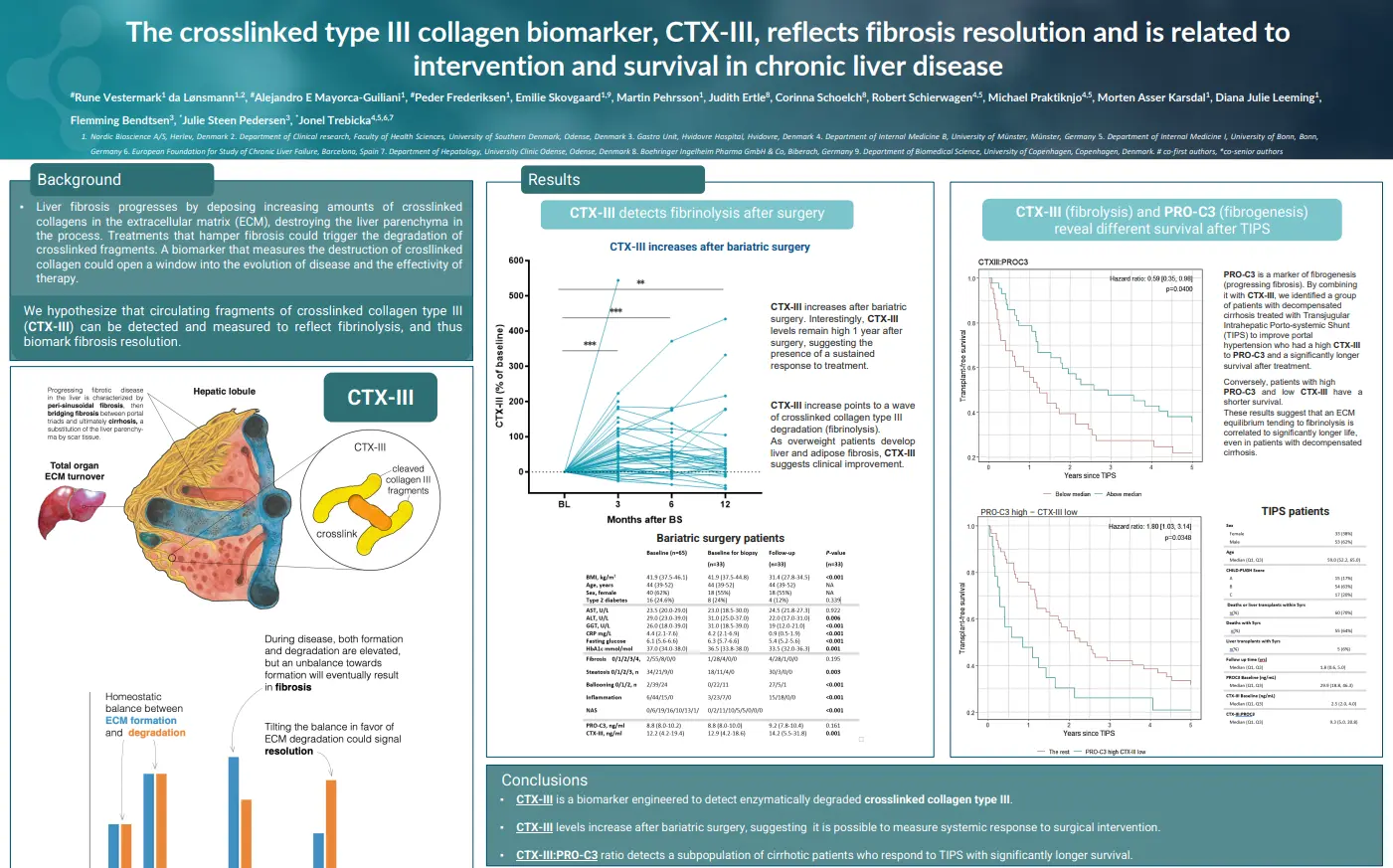
C3F levels are elevated in patients with stenotic Crohn’s disease (CD) compared to those with luminal CD at baseline. In the stenotic group, C3F levels rise further following surgical resection compared to baseline, 1 month, 3 months, and 6 months post-surgery, suggesting that C3F reflects fibroblast activity during fibrostenosis and tissue remodeling following surgery. While C3F demonstrates only modest discriminatory ability across comparisons (AUC 0.65–0.69), its positive correlation with CRP supports an association with inflammation leading to collagen deposition and fibrosis in stenotic CD. Additionally, a negative correlation with disease duration suggests that C3F may reflect fibrosis at earlier stages of the disease.
In luminal CD patients, C3F levels were significantly higher at 12 months than at baseline and 6 months. Moreover, early C3F levels (baseline, 3 months, and 6 months) were significantly associated with ileal stenosis observed at 12 months, suggesting that elevated C3F may predict future endoscopic disease activity or potential relapse. Overall, these findings underscore the potential of C3F as a novel biomarker for intestinal fibrosis in Crohn’s disease.