Primary sclerosing cholangitis (PSC) is a chronic liver disease that leads to a damage of the bile duct, inflammation and fibrosis.
As there is a need for non-invasive biomarkers that can accurately assess the disease severity and prognosis, this study evaluated the ability of extracellular matrix remodeling markers to diagnose fibrosis stage and predict PSC-related fibrosis progression and clinical events.
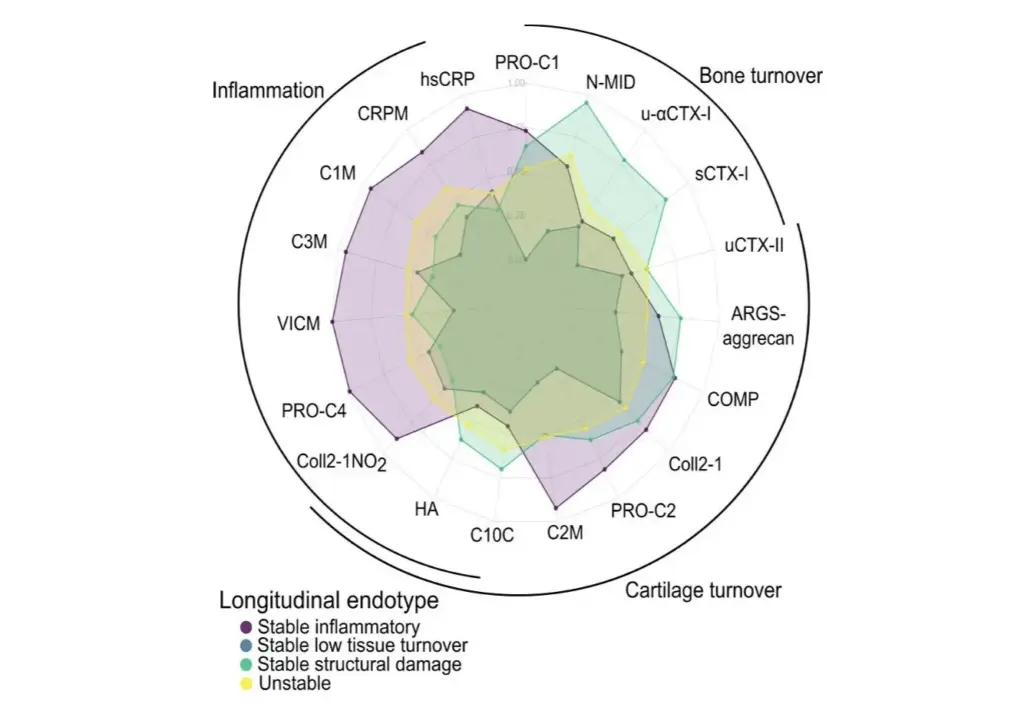
Our findings in this study showed that nordicPRO-C3™ correlated with fibrosis stage, and nordicPRO-C3™ and ELF score provided discrimination of advanced fibrosis and cirrhosis and predicted PSC-related events and fibrosis progression. Therefore, nordicPRO-C3™ and ELF may be utilized for staging and can act as prognostic markers in PSC, contributing to improving current screening and treatment of immune-mediated liver diseases.
Article: Serologic extracellular matrix remodeling markers are related to fibrosis stage and prognosis in a phase 2b trial of simtuzumab in patients with primary sclerosing cholangitis