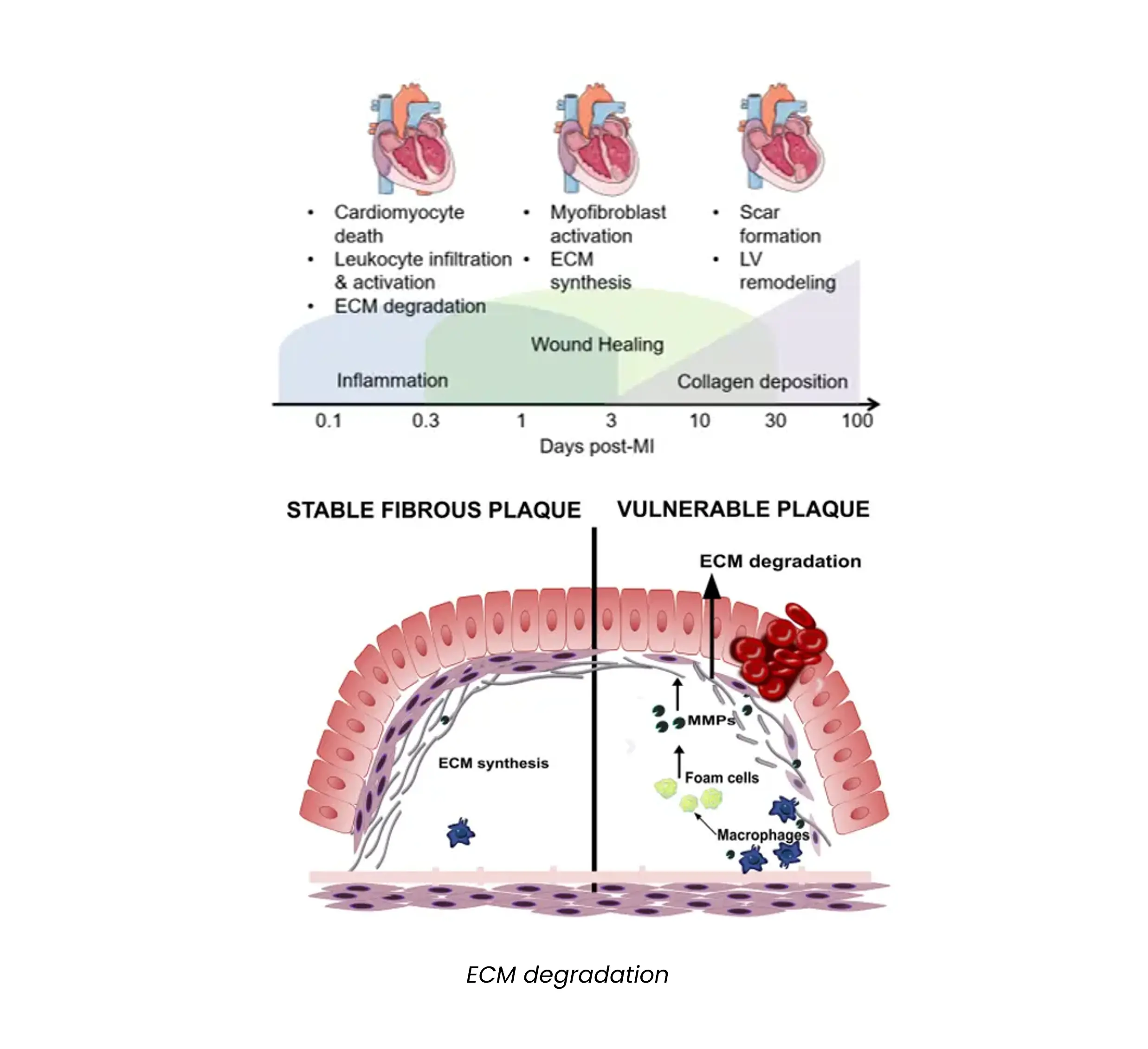
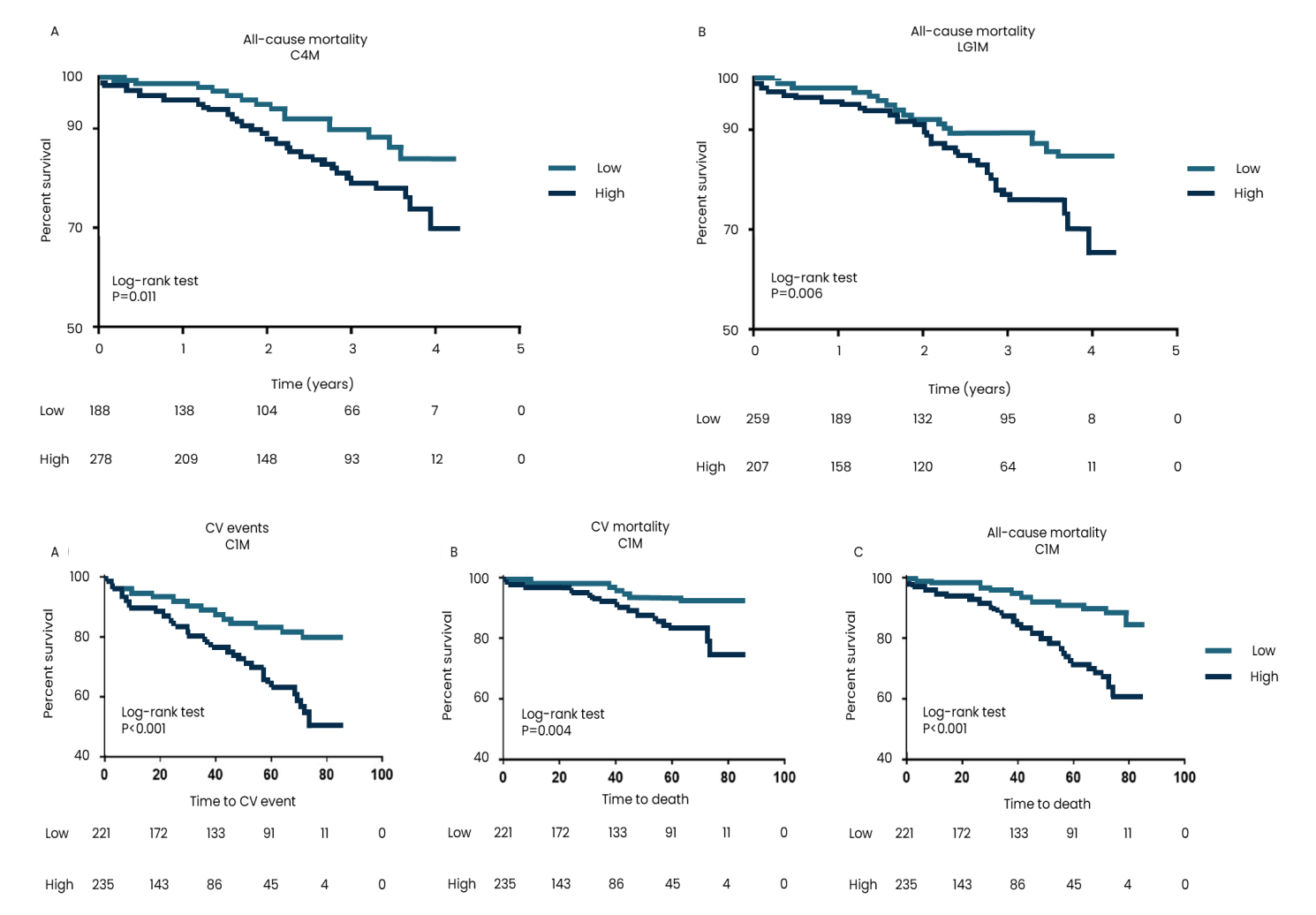
Atherosclerosis is commonly assessed by computed tomography angiography. Cardiac magnetic resonance imaging is also frequently being implemented in diagnosis and risk assessment of plaque rupture.
Atherosclerosis can also be assessed invasively by angiography but is not recommended as a screening tool. Blood-based biomarkers are also implemented in diagnosis, such as C-reactive protein (CRP), lipids and lipoproteins.
Patients experiencing MI often complain about chest pain and discomfort in the chest, shoulders, and arms. Patients can also experience shortness of breath, nausea, and feeling faint. However, these symptoms are not unique to cardiovascular diseases, and further diagnostic measures are applied.
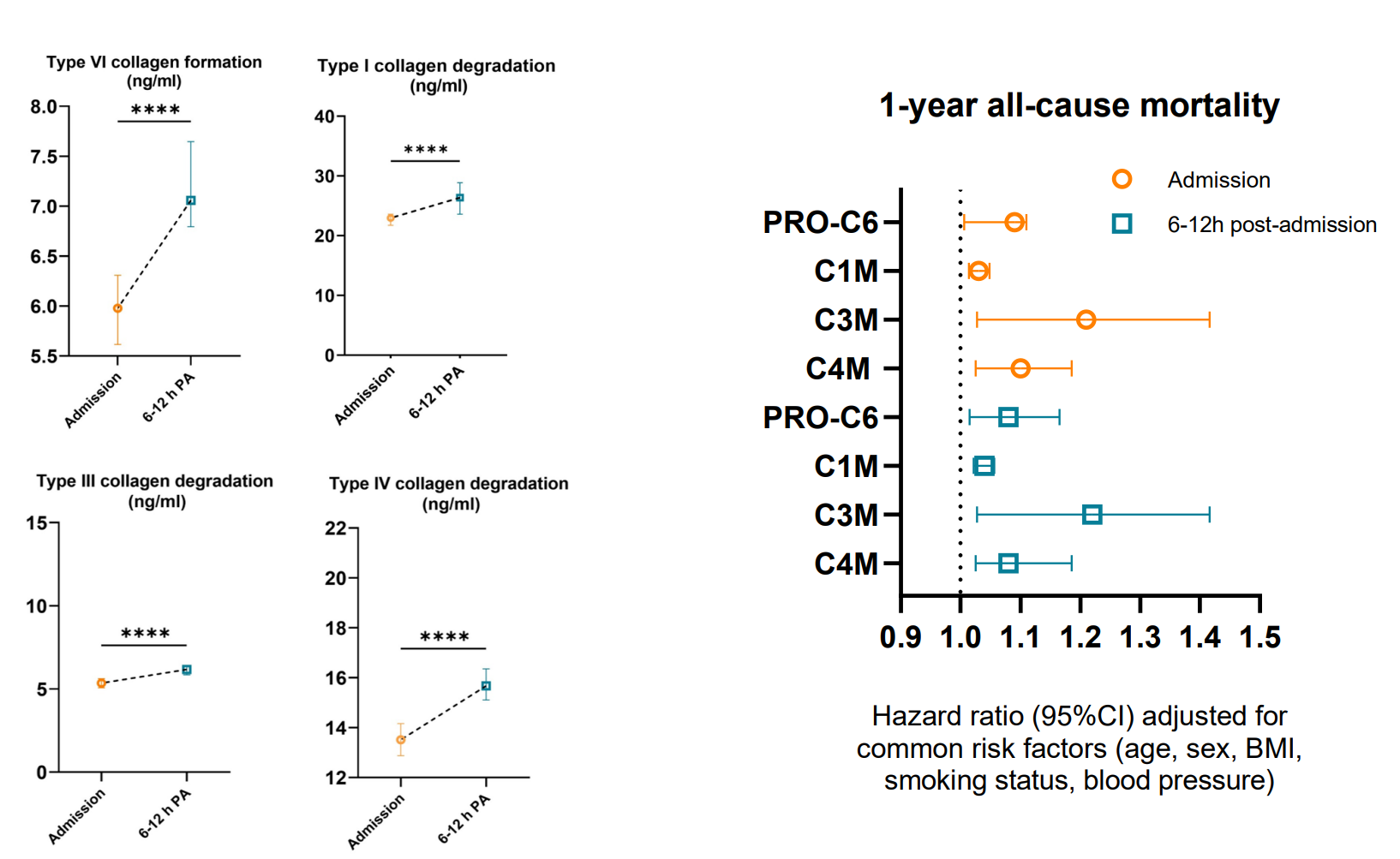
Because MI leads to tissue damage, cardiac-specific biomarkers are released such as troponin, and can be measured in the blood of patients.
Electrocardiogram (ECG) is the most common non-invasive tool used for diagnosis of MI and it is often used repeatedly over minutes to hours to monitor changes in the cardiac rhythm after admission to hospital.
ECG is employed to separate a MI with ST-segment elevation myocardial infarction (STEMI) and a MI with non-ST-segment elevation myocardial infarction (NSTEMI). Imaging techniques, such as angiography, are used to assess the blockage of the coronary arteries.