Chronic kidney disease (CKD) is a long-term condition where the kidneys gradually lose function over time, often without noticeable symptoms in the early stages. CKD affects over 800 million people worldwide and is a major driver of morbidity, mortality, and healthcare costs. Early identification of patients at risk of rapid progression is essential to improve outcomes and tailor therapeutic interventions.
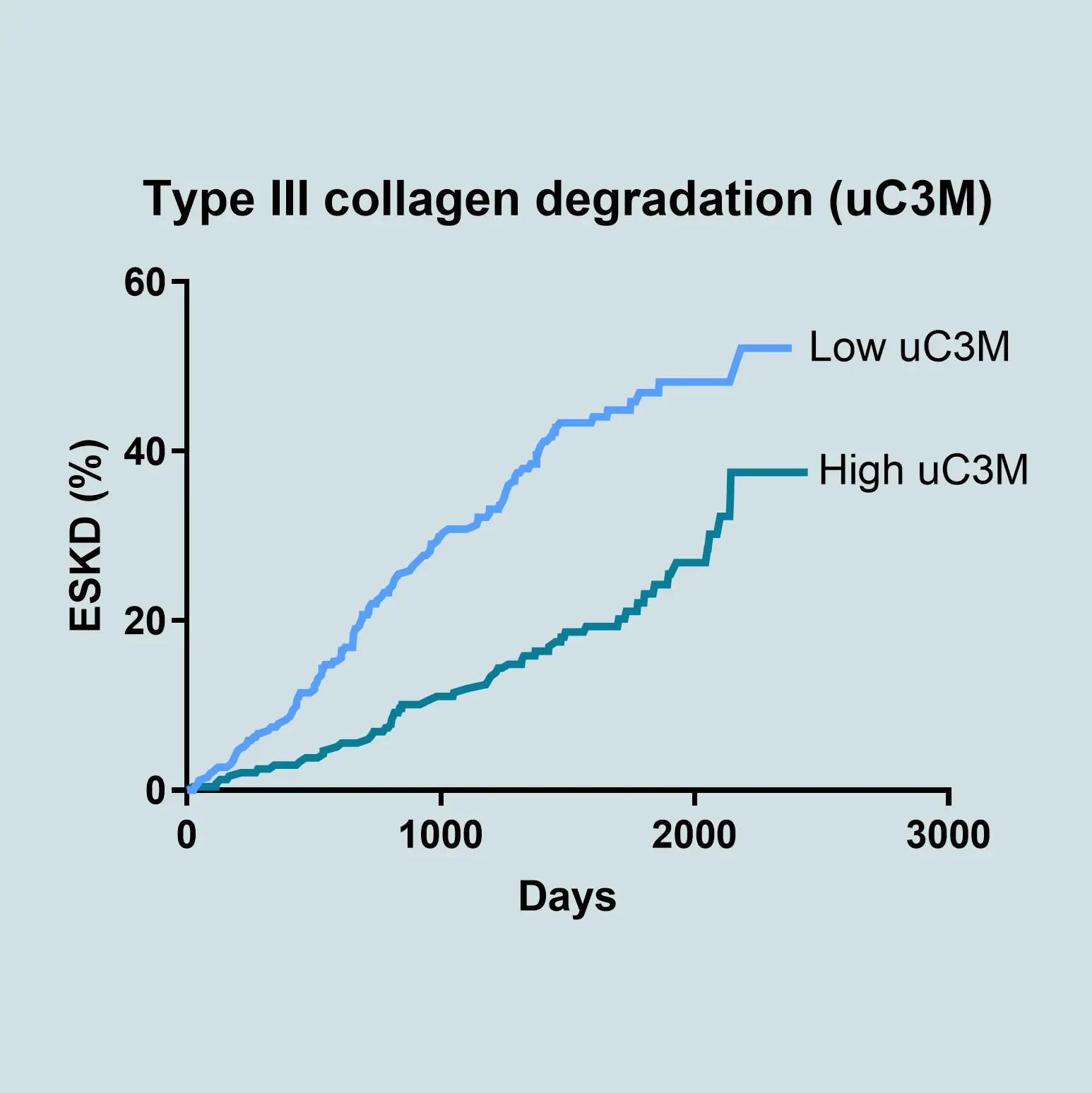
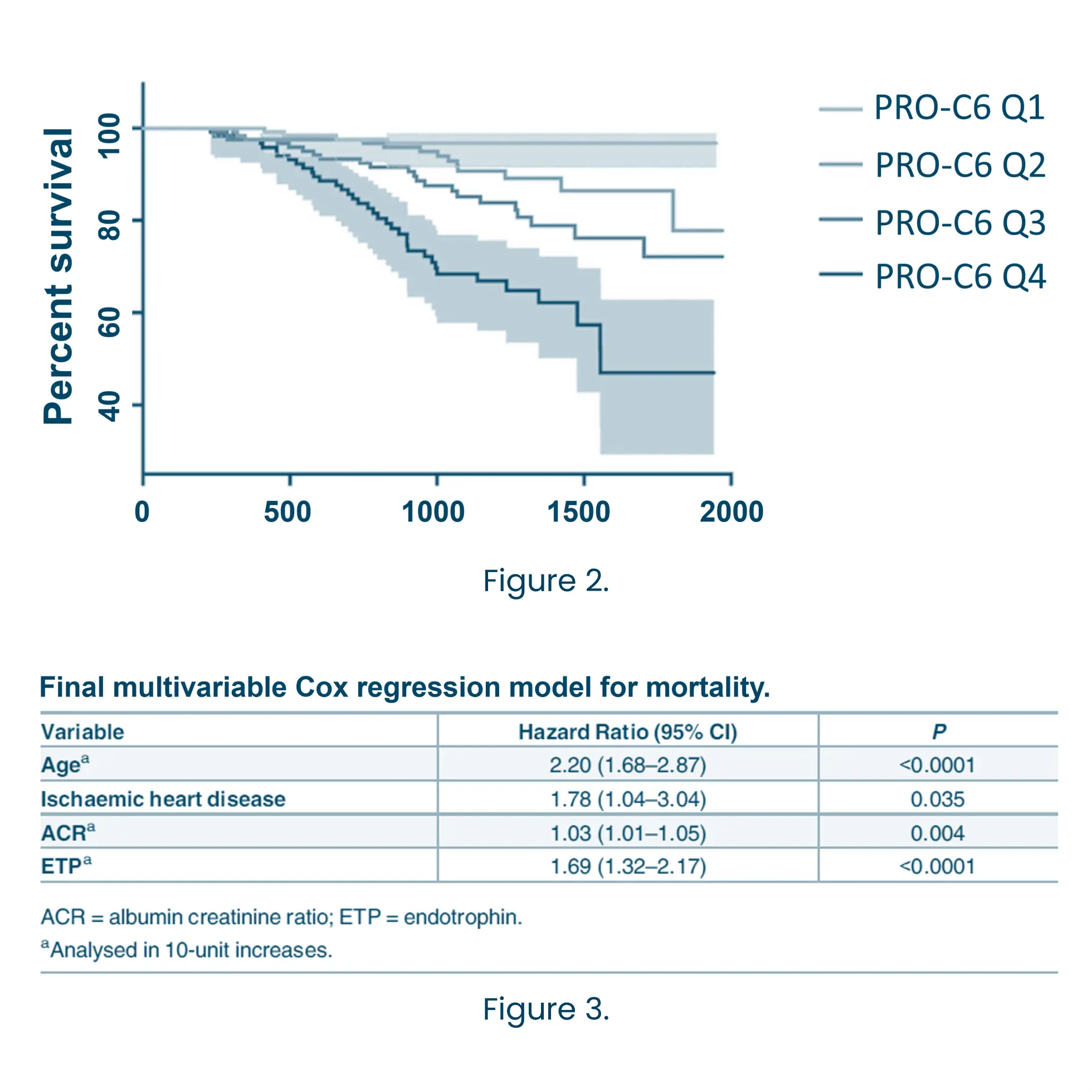
The most widely used and accepted clinical markers to predict the progression of CKD are eGFR and albuminuria, but they often fail to detect early or dynamic changes in disease activity. Biomarkers offer a powerful solution by providing mechanistic insights into underlying pathophysiology. Biomarkers related to the extracellular matrix (ECM) reflect ongoing tissue remodeling and fibrosis—key processes driving CKD progression. ECM biomarkers can predict disease trajectory more accurately than conventional measures alone.