Endotrophin has a receptor now: CD44-STAT3 as the Endotrophin signaling route in cancer and fibrosis
March 4, 2026
For years, Endotrophin has been discussed as a circulating signal linked to fibroinflammatory disease severity and adverse outcomes. But there has been a persistent gap between association and a clearly defined, targetable mechanism. Now, a new report addresses that gap by identifying CD44 as a receptor for Endotrophin and connecting Endotrophin–CD44 binding to STAT3 signaling in hepatocellular carcinoma (HCC).
This publication anchors that association mechanistically by showing that Endotrophin is produced in fibrotic liver by COL6A3‑rich hepatic stellate cells (HSCs) and is not merely a degradation product, but a bioactive hormone-like peptide that signals into neighboring cells.
In the study’s experimental system, Endotrophin binding to CD44 activated STAT3 signaling and was linked to phenotypes relevant to tumor progression, including epithelial–mesenchymal transition (EMT), proliferation, and sorafenib resistance.
Fibroblast-derived Endotrophin signals into CD44+ cells and sustains itself
The paper further describes that hepatic stellate cell–derived Endotrophin targets pericentral CD44+ tumor cells, induces COL6A3 expression, and sustains Endotrophin production via a STAT3-dependent feedback loop.
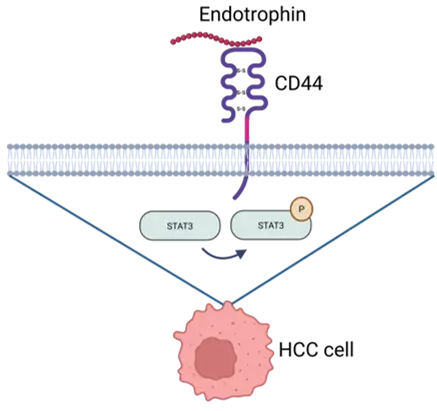
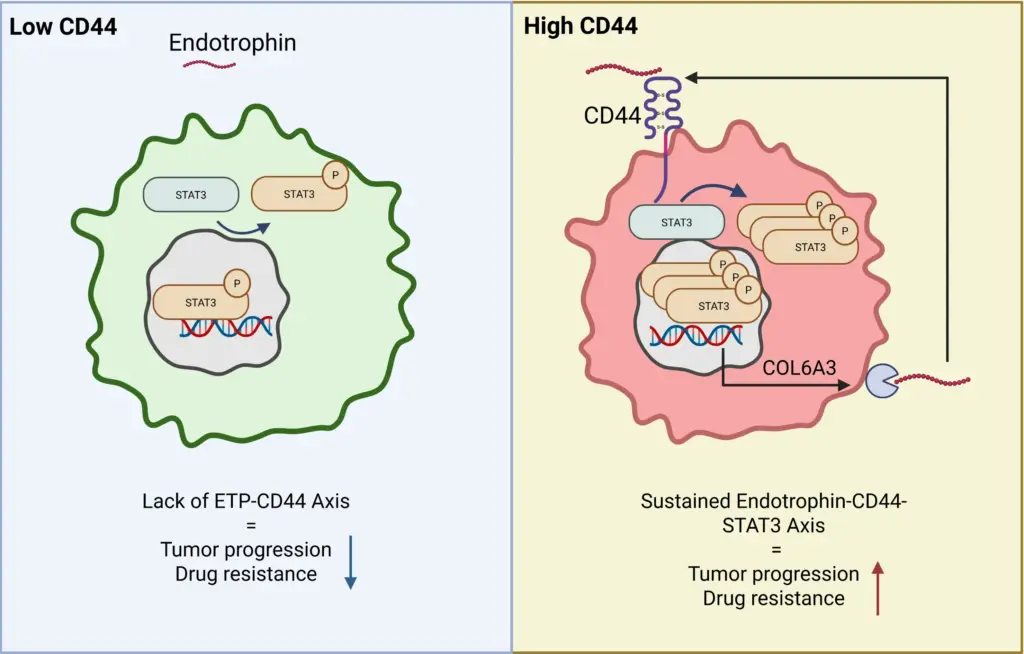
By defining CD44–STAT3 as the principal Endotrophin signaling route in HCC, the paper suggests a shared signaling logic by which Endotrophin‑rich fibrotic niches (kidney, heart, adipose tissue) can drive organ damage and risk of outcome through similar downstream pathways.
Interested in how Endotrophin relate to fibroblast activity?
Webinar:
Collagen VI and Endotrophin Biomarkers for Mechanism, Prognosis, and Drug Development
Circulating Endotrophin likely reflects an activated Endotrophin-CD44–STAT3 loop in fibrotic organs. High circulating Endotrophin is not only a marker of fibroblast activity but a surrogate for an ongoing pathogenic signaling circuit. Inhibiting the Endotrophin–CD44–STAT3 axis (genetic Col6a3/Cd44 deletion, STAT3 inhibition, or CD44‑binding‑defective Endotrophin mutants) attenuates fibrosis, EMT, steatosis and chemoresistance, positioning this axis as a therapeutic target in metabolic dysfunction-associated HCC.
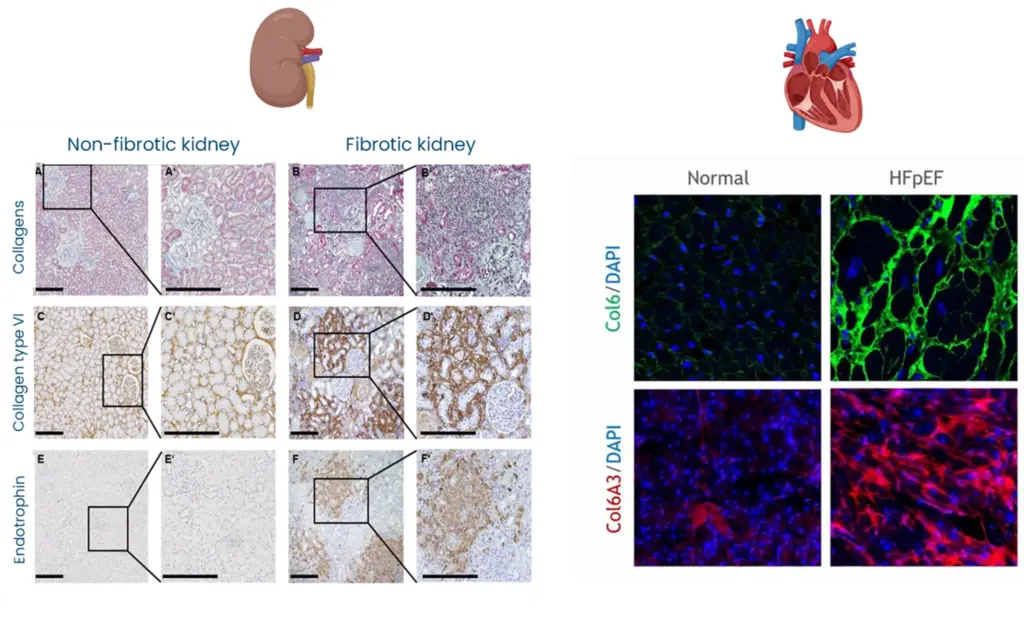
Endotrophin does not only stage and prognosticate fibrotic disease but can also identify patients in whom Endotrophin‑driven fibro-inflammatory signaling is active and may be actionable by Endotrophin‑neutralizing antibodies or STAT3‑targeted interventions.
Why measure outcomes and risk signals across chronic disease
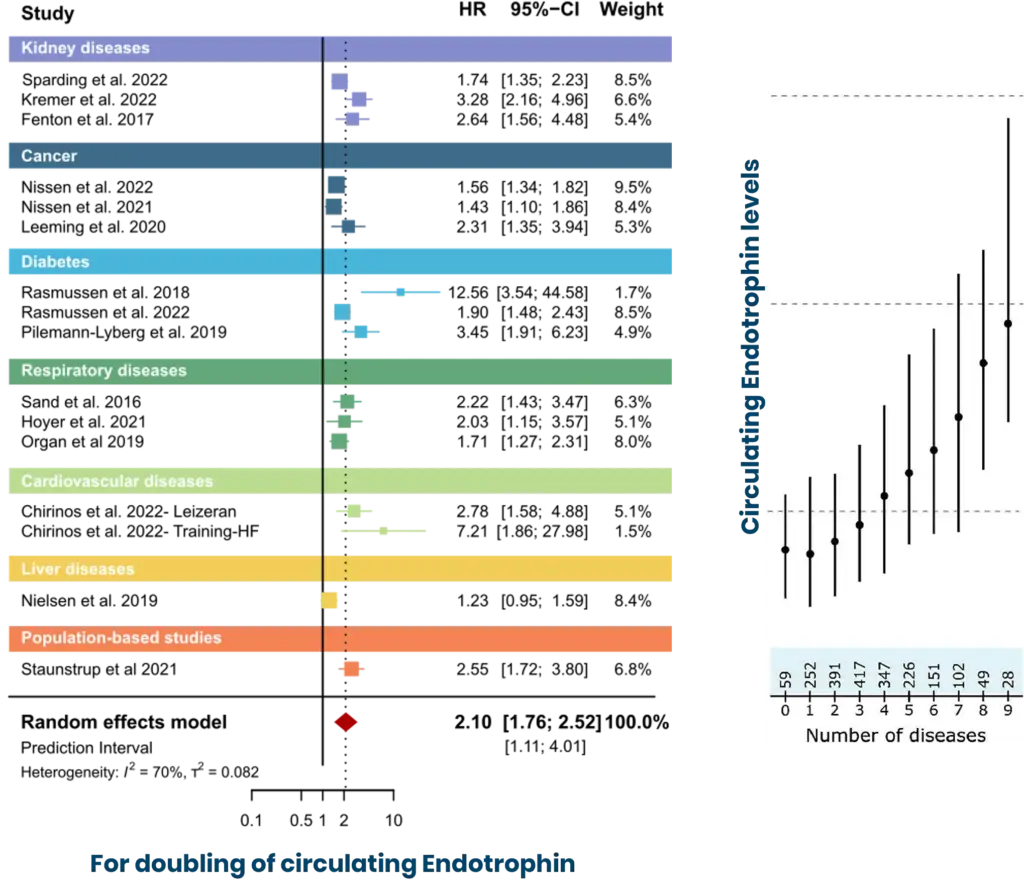
Endotrophin in the blood reflects fibro-inflammatory remodeling associates with cardiac and kidney fibrosis, mortality, and progression of MASH, cirrhosis, and HCC, with levels rising across the spectrum of metabolic diseases.
Endotrophin is not just “another fibrosis marker,” but a circulating readout of active fibro-inflammatory biology. In a systematic review and individual participant data meta-analysis, higher circulating Endotrophin independently associated with increased mortality risk across a broad range of chronic diseases: a core clinical reason to measure Endotrophin and nordicPRO-C6™ in programs where baseline biology and outcome risk need to be quantified.
Mechanistically, Endotrophin upregulates and releases into circulation as a consequence of recurring pathophysiological mechanisms across diseases. The functional implication of circulating Endotrophin supports PRO-C6 as an operational biomarker in clinical programs, where a blood-based measurement aligned with fibroblast-driven remodeling biology can be used at scale and longitudinally.
How does CD44 connect to existing literature on Endotrophin?
The new receptor paper changes how to interpret associations by providing a defined signaling handle. CD44 was identified as an Endotrophin receptor, and Endotrophin–CD44 engagement activated STAT3 signaling with disease-relevant phenotypes. When a circulating molecule is both outcome-associated and connected to a receptor axis, measuring Endotrophin and PRO-C6 becomes a practical component of biology-aware stratification and monitoring in fibro-inflammatory clinical programs. Nordic’s measurement approach centers on assays that quantify Endotrophin-related biology in circulation, including PRO-C6-based measurement supported through clinical evidence.
If your program involves fibro-inflammatory biology, position Endotrophin measurement as an evidence-aligned readout to support:
- Baseline characterization of fibroblast-associated signaling burden,
- Risk stratification informed by published outcome associations across chronic disease,
- Mechanistic hypothesis testing in settings where CD44/STAT3 biology is part of the rationale.



